
خدمات
الإستشارات والتدريب

التميٌّز المؤسسي
جائزة قطر للتميز الحكومي ونموذج التميز المؤسسي الأوروبي EFQM

دليل السياسات
يشمل دليل السياسات والإجراءات تصنيف الأقسام والوحدات الإدارية وتحليل العمليات وتوثيقها واعتمادها

التخطيط الإستراتيجي
بناء الخطط الإستراتيجية ومتابعتها وفقا لرؤية قطر الوطنية 2030

التدقيق الداخلي
النتائج والتوصيات وتحليل بهدف تصحيح حالات عدم المطابقة وتحسين الأداء في مختلف الأقسام والإدارات

معايير الأيزو المختلفة
التأهيل لشهادات ضبط الجودة العالمية ايزو ISO وغيرها من المعايير العالمية

إدارة المخاطر
بناء سجل إدارة المخاطر ومعالجتها من خلال نهج مركزي كليّ وليس بشكل مستقل داخل الوحدات
Magnus Management – Quality, Strategy, EFQM Excellence


ماجنس مانجمنت للإستشارات ذ.م.م
عن الشركة؟
ماجنس للإستشارات شركة استشارات إدارية تم تأسيسها عام 2017 في الدوحة - قطر، لتقديم خدمات الإستشارات والتدريب في مجال التخطيط الإستراتيجي والتميز المؤسسي ومعايير الأيزو المختلفة. بالإضافة إلى إدارة المخاطر والتدقيق الداخلي والعديد من الخدمات الإدارية والتطوير المختلفة التي تستهدف مساعدة الشركات في تطوير اعمالها وتبسيطها وتحقيق التميز المؤسسي وفق معايير ومتطلبات توجهات دولة قطر وزيادة فعالية العمليات وزيادة الإنتاجية.
إن السـعي المسـتمر لتطويـر القطـاع الحكومـي فـي دولـة قطـر لمواكبـة التسـارع الـذي يشـهده العالـم فـي المنافسـة علــى التحــول لإقتصــاد المعرفــة يحتــم علــى الجهــات الحكوميــة والخاصة الســير قدمــا لتقديــم خدمــات ذات جــودة عاليــة للمتعامليــن، والعمــل علــى تطويرهــا ورفــع مســتويات كفاءتهــا وفعاليتهــا فــي ظــل تزايــد الطلــب علــى الخدمــات الحكوميــة مــن كافــة شــرائح المجتمــع القطــري واختــلاف احتياجاتهــم وارتفــاع مســتويات توقعاتهــم.
إن الإلتزام بمعايير الأيزو يعني وجود منهجية علمية في عملية اتخاذ القرار وإيجاد ثقافة التحسين المستمر في المؤسسة، الأمر الذي يسهم في تعزيز جودة المنتجات والخدمات من خلال وضع معايير وإرشادات تساعد الشركات على تطبيق ممارسات أفضل في تصميم وتصنيع المنتجات وتقديم الخدمات. وتحسين رضا العملاء من خلال تقديم منتجات ذات جودة مضمونة وخدمات موثوقة وملتزمة بالمعايير الدولية.
Magnus Management – Quality, Strategy, EFQM Excellence


رؤيتنا
أن نسهم في دعم المؤسسات القطرية والعربية وتحقيق الاكتفاء العربي في مجال الإستشارت الإدارية
خدمات الاستشارات والتدريب
تقدم ماجنس مانجمنت للإستشارات ذ.م.م خدمات الإستشارات الإدارية لمختلف المؤسسات والهيئات الحكوميةوالخاصة بهدف تطبيق أعلى معايير الجودة والتطوير والتميز في الأداء والإلتزام بأفضل الممارسات العالمية، الأمر الي سينعكس على التميز في الأعمال والخدمات والحصول على جودة عالية ونسبة رضى عملاء مرتفعة
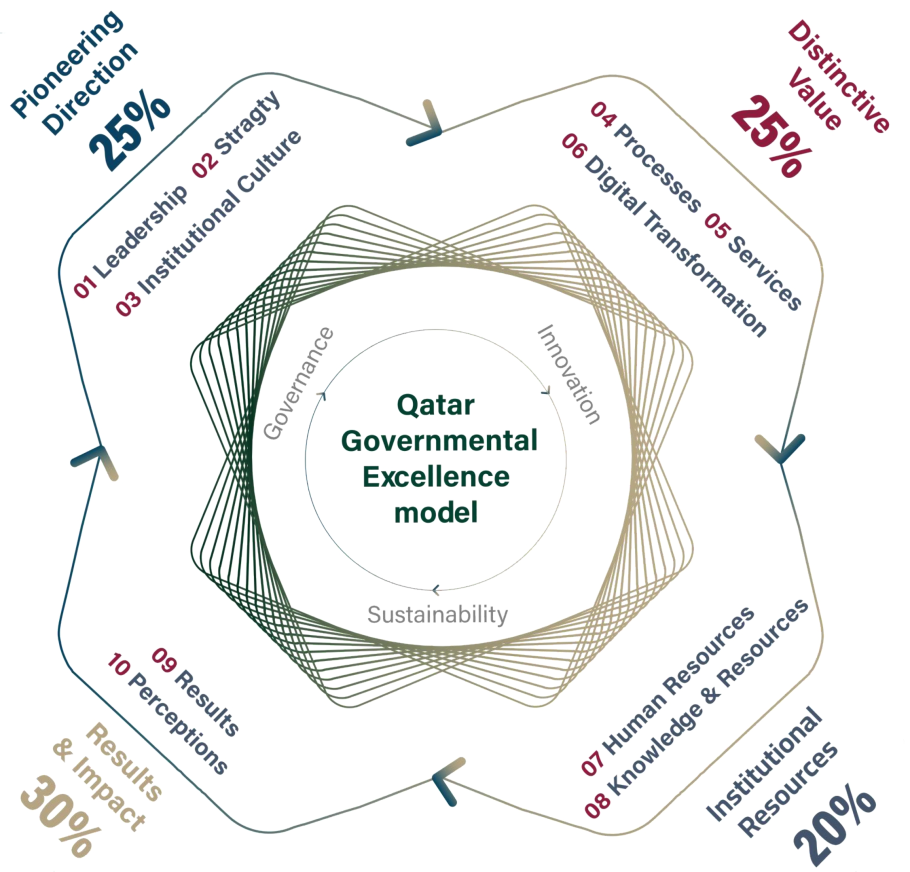
جائزة قطر للتميُّز المؤسسي ونموذج التميُّز الأوروبي EFQM Excellence Model 2022
جاء القرار رقم (63) لسنة 2023، الذي أصدره حضرة صاحب السمو الشيخ تميم بن حمد آل ثاني أمير البلاد المفدى، بإنشاء جائزة قطر للتميز الحكومي، ليدعم توجه الدولة وإستراتيجيتها للارتقاء بمستوى جودة الأداء الحكومي، وتشجيع الجهات الحكومية والموظفين على التنافس في تطبيق أعلى معايير الجودة والتطوير والتميز في الأداء، من خلال تكريم الإنجازات والجهود المتميزة لجميع الفئات المساهمة في تحقيق الرؤية الوطنية لدولة قطر.
تحديد الأهداف الرئيسية لمؤسسة أو منظمة وتحديد السبل والخطط التي ستسلكها لتحقيق تلك الأهداف. يتضمن هذا العمل تحليلاً شاملاً للبيئة الداخلية والخارجية للمؤسسة، بما في ذلك فحص القدرات والضعف الداخلية والفرص والتحديات الخارجية. يهدف التخطيط الاستراتيجي إلى تحديد الاتجاهات الرئيسية وتحديد السياسات والإجراءات التي يجب اتخاذها لضمان تحقيق الأهداف المحددة. تعتبر الخطط الاستراتيجية أداة هامة لتحديد اتجاه المؤسسة وتحديد كيفية استخدام الموارد بكفاءة لتحقيق أهدافها على المدى الطويل.
بطاقات الوصف الوظيفي تعتبر وسيلة فعّالة لتوضيح الواجبات والمسؤوليات الرئيسية المرتبطة بوظيفة معينة. يمكن استخدام هذه البطاقات لتزويد الموظفين بفهم واضح حول ما يتوقع منهم في دورهم الوظيفي، حيث يتم بناء الهيكل الوظيفي وبطاقات الوصف الوظيفي والأهداف الوظيفية لمساعدة المنظمة على تحقيق أهدافها بكفاءة وفاعلية وتحقيق الإنسجام بين مختلف الإدارات والأقسام والأنشطة، وتفادي التداخل والإزدواجية والإختلافات وغيرها.
وضع مؤشرات قياس الأداء s'KPI لضمان الرقابة والمتابعة المستمرة للنتائج المحققة من الأهداف والتأكد من تحقيق المعدلات المستهدفة مثل معدل الرضا – الجودة – الإنتاجية والتأكد من فعالية الخطة والأهداف والمهام.
بناء سجل إدارة المخاطر الهادف إلى معالجة المخاطر من خلال نهج مركزي كلي، وليس بشكل مستقّل داخل الوظائف أو الوحدات. كما تتيح معالجة المخاطر الفرصة للموظفين لفهم كيفية تأثير تصرفاتهم الفردية عىل أهدافهم وأهداف المؤسسة الإستراتيجية بُرّمتها بناء على متطلبات ديوان المحاسبة.
نقوم بتطبيق معايير الأيزو وتأهيل عملائنا للحصول على شهادة الأيزو في قطر، حيث يمر المشروع بعدة مراحل بدءا بتحليل الفجوات بين النظام الحالي ونظام الأيزو، وتدريب الموظفين وتعريفهم بمعايير الأيزو وأهميتها، ومن ثم تبدأ مرحلة التطبيق لهذه المعايير. بعد ذلك نقوم بعملية التدقيق الداخلي للحرص على الامتثال للمعايير والتأهل للحصول على شهادة الايزو. وتعتبر معايير الأيزو مصممة لتحسين جودة المنتجات والخدمات من خلال تحديد متطلبات وعمليات محددة، يمكن للمؤسسات تحسين إنتاجيتها وضمان تقديم منتجات أو خدمات ذات جودة عالية ومنها:
ISO 9001:2015 - نظام إدارة الجودة - السياسات والعمليات والإجراءات اللازمة لتخطيط وتنفيذ وتقديم (المنتجات / الخدمات) بهدف التأكد من قدرة المنشأة على الوفاء بمتطلبات العملاء، ويعمل نظام إدارة الجودة على ترسيخ مبدأ التحسين المستمر للخدمات والمنتجات المقدمة
ISO 14001:2015 - نظام إدارة البيئة - معيار دولي يركز على إدارة الأثر البيئي للمؤسسات. الهدف من هذا المعيار هو توجيه المؤسسات في تحسين أدائها البيئي والتحكم في التأثير البيئي
ISO 45001:2018 - نظام إدارة الصحة والسلامة المهنية - هو معيار دولي يركز على تحسين السلامة والصحة المهنية في مكان العمل. هذا المعيار يهدف إلى توفير إطار لتحسين أداء نظام إدارة الصحة والسلامة في المؤسسات
ISO 27001:2013 - نظام إدارة أمان المعلومات - معيار دولي يركز على حماية المعلومات داخل المؤسسات. يهدف هذا المعيار إلى توفير إطار لتأمين المعلومات وتقديم إرشادات لتحسين نظم إدارة أمان المعلومات
ISO 31000:2018 - إدارة المخاطر - معيار دولي يتعلق بإدارة المخاطر. يُعتبر هذا المعيار إطارًا لإدارة المخاطر ويهدف إلى توفير توجيهات شاملة تساعد المؤسسات على فهم وتطبيق مفاهيم إدارة المخاطر بشكل فعّال. يشمل المعيار عناصر أساسية مثل تحديد وتقييم المخاطر، وتحديد السياق الذي يؤثر على إدارة المخاطر، وتطبيق استراتيجيات للتعامل مع المخاطر بطريقة فعالة ومستدامة. يشجع ISO 31000 على تكامل إدارة المخاطر في هياكل الحوكمة والعمليات التنظيمية.
وغيرها من المعايير العالمية
ضمان أن نظام إدارة الجودة في المنظمة يعمل بفعالية ويتوافق مع متطلبات المعيار. حيث تتم عملية التدقيق الداخلي من خلال تقييم ومراجعة لنظام إدارة الجودة الخاص بالمنظمة، وهو يتضمن مراجعة وتقييم العمليات والإجراءات والتوثيق والأداء العام وقياس أداء العمليات وتحديد المجالات التي يمكن تحسينها وأن الموارد تستخدم بشكل صحيح وتقديم توصيات لتحسين العمليات والأداء
بناء دليل السياسات والإجراءات بما يشتمل على تصنيف الأقسام والوحجات الإدارية وتحليل العمليات وتوثيقها وتحديد مصفوفة الصلاحيات
جائزة قطر للتميز الحكومي
أطلقت جائزةُ قطر للتميز الحكومي في أغسطس 2023، وذلك بهدف الارتقاء بمُستوى جودة الأداء الحكوميّ وتشجيع الجهات الحكومية والموظّفين على التنافس في تطبيق الجودة والتطوير والتميز في الأداء، من خلال تكريم الإنجازات والجهود المتميزة لجميع الفئات المساهمة في تحقيق رؤية قطر 2030
التوجه الريادي
القيادة، الإستراتيجية والثقافة المؤسسية
القيمة النوعية
العمليات، الخدمات والتحول الرقمي
الموارد المؤسسية
الموارد البشرية، الموارد والمعرفة
النتائج والأثر
النتائج والانطباعات

معرض صنع في قطر 2023
قامت ماجنس مانجمنت للإستشارات ذ.م.م بعقد ندوة تعريفية عن التميز المؤسسي والجودة وجائزة قطر للتميز الحكومي خلال مشاركتنا في معرض صنع في قطر لعام 2023 في الدوحة تم خلال الندوة عرض محاور جائزة قطر للتميز الحكومي ومعاييرها ومناقشة أهمية الالتزام بمعايير الأيزو العالمية، ومناقشة أهم مواصفات الأيزو التي تتسابق مختلف الشركات للحصول عليها ودورنا في تقديم خدمات الإستشارات والتدريب لتأهيل عملائنا في الامتثال لها وتأهيلهم للحصول على شهادة الأيزو. وتم القيام بمناقشات تعريفية مع الزوار بهدف التوعية بأهمية معايير الأيزو والجودة كمعايير ال ISO 9001:2015 QMS, ISO 14001:2015 EMS,ISO45001:2018 OH&S



شهاداتنا
عملائنا السعداء
سعدنا بالعمل مع العديد من المؤسسات والهيئات الحكومية والخاصة وتقديم خدمات الإستشارات والتدريب في مجال التخطيط الإستراتيجي والتميز المؤسسي ومعايير الأيزو المختلفة. بالإضافة إلى إدارة المخاطر والتدقيق الداخلي والعديد من الخدمات الإدارية والتطوير المختلفة.






خبراتنا
نحن نمتلك
استشاريون معتمدون
مقيم من EFQM
نموذج التميز الأوروبي
المنظمة العالمية للتخطيط الاستراتيجي
الوحيدون في قطر
مدققون معتمدون لأنظمة الأيزو
لأنظمة ISO 9001, ISOO 14001, ISO 45001 & ISO 27001
مستعد؟ أبدأ رحلة تطوير أعمالك
نحن هنا لبدء مشروعك الجديد والانتهاء منه قريبًا
- اتصل بنا الآن97431009655+
- البريد الإلكتروني info@mmcc.qa
- عنوانناالطريق الدائري الرابع، مبنى ريجس للأعمال، الطابق الأول، مكتب 133
